Optimizing the Haber Process: Designing for Maximum Ammonia Yield
NGSS Alignment
- Performance Expectation: HS-PS1-6: Refine the design of a chemical system by specifying a change in conditions that would produce increased amounts of products at equilibrium.
- Evidence Statement(s): Refine a solution to a complex realworld problem, based on scientific knowledge, student-generated sources of evidence, prioritized criteria, and tradeoff considerations.
- Science and Engineering Practices (SEP): Constructing Explanations and Designing Solutions
- Disciplinary Core Ideas (DCI): PS1.B: Chemical Reactions; ETS1.C: Optimizing the Design Solution
- Crosscutting Concepts (CCC): Stability and Change
Overview for Educators
Duration: Approximately 45 minutes Prerequisites: Students should understand the basic concept of reversible reactions and have a foundation in collision theory. How it Works: This interactive simulation allows students to manipulate the concentration, pressure, and temperature of a closed system containing nitrogen, hydrogen, and ammonia. By observing the macroscopic graphs and particulate-level collisions, students deduce Le Chatelier’s Principle and apply it to a real-world chemical engineering problem: maximizing ammonia yield in the Haber Process.
Introduction
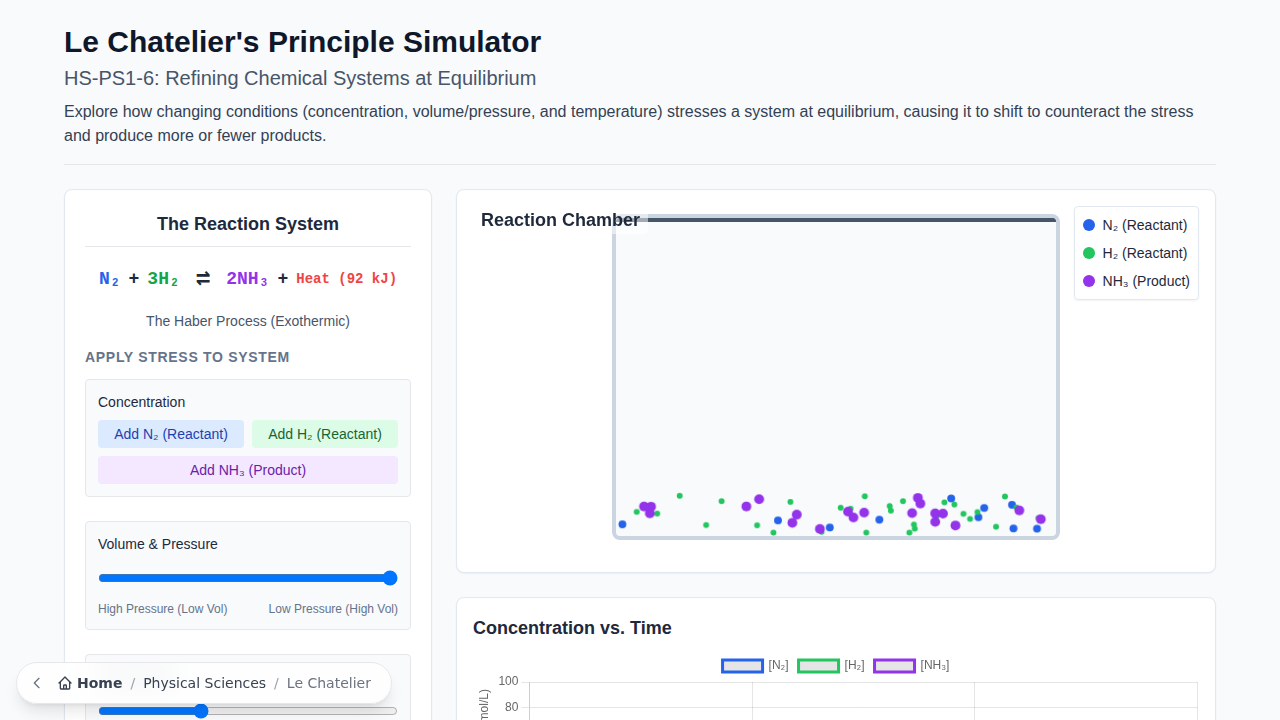
Ammonia (NH3) is one of the most highly produced inorganic chemicals globally. It is essential for the manufacturing of fertilizers, which support a significant portion of the world’s food production. The industrial synthesis of ammonia, known as the Haber Process, combines nitrogen (N2) and hydrogen (H2) gases in a reversible, exothermic reaction: N2(g) + 3H2(g) ⇌ 2NH3(g) + Heat (92 kJ)
As a chemical engineer working for a major agricultural chemical company, your task is to maximize the yield of ammonia. Because the reaction is reversible, the system reaches an equilibrium state where the rate of the forward reaction equals the rate of the reverse reaction. You must use Le Chatelier’s Principle to determine the optimal conditions for production.
Real-World Trade-offs: When designing an industrial plant for the Haber Process, engineers face a classic dilemma balancing kinetics vs. thermodynamics. A low temperature favors the production of ammonia (thermodynamics), but causes the reaction to run too slowly (kinetics). A high pressure favors ammonia production, but the equipment required to safely maintain extreme pressure is very expensive.
Access the simulation here: Le Chatelier’s Principle Simulator
Task Instructions
Part 1: Initial Exploration
Open the simulation. The system is initially at equilibrium.
- Observe the macroscopic view (concentrations) and the particulate view (molecules in the chamber).
- Identify the reactants and products of the Haber Process.
Part 2: Analyzing the Effect of Concentration Changes
Your company has an excess supply of hydrogen gas.
- In the simulation, inject more H₂ gas into the system.
- Observation: What happens to the concentration of NH₃ as the system re-establishes equilibrium? What happens to the concentration of N₂?
- Explanation: Use Le Chatelier’s Principle to explain why this shift occurs. Describe what is happening at the molecular level (e.g., collisions between particles).
- Application: If your goal is to maximize NH₃ production, how should you manage the concentrations of N₂ and H₂ continuously?
Part 3: Analyzing the Effect of Temperature Changes
The Haber Process is an exothermic reaction, meaning it releases heat (92 kJ).
- Reset the simulation.
- Increase the temperature of the system.
- Observation: What happens to the concentration of NH₃?
- Decrease the temperature of the system.
- Observation: What happens to the concentration of NH₃?
- Explanation: Use Le Chatelier’s Principle to explain how temperature changes affect an exothermic equilibrium system. Treat heat as a “product” in the reaction equation.
- Application: Based strictly on equilibrium principles, would you want to run this reaction at a high temperature or a low temperature to maximize NH₃ yield?
Part 4: Analyzing the Effect of Volume/Pressure Changes
The reaction involves gases: 1 mole of N₂ and 3 moles of H₂ react to form 2 moles of NH₃.
- Reset the simulation.
- Decrease the volume of the reaction chamber (which increases the pressure).
- Observation: What happens to the concentration of NH₃?
- Explanation: Use Le Chatelier’s Principle to explain how changing the volume/pressure affects this specific equilibrium system. Consider the number of moles of gas on the reactant and product sides.
- Application: To maximize NH₃ yield, should the reaction be carried out at high pressure or low pressure?
Part 5: Refining the Design (Engineering Trade-offs)
You have determined the theoretical conditions to maximize ammonia yield. Now, consider the realworld engineering constraints:
- Temperature Trade-off: While a low temperature favors the formation of ammonia (thermodynamics), at very low temperatures, the reaction rate is too slow to be economically viable (kinetics).
- Pressure Trade-off: High pressure favors ammonia production, but building and maintaining high-pressure reaction vessels is expensive and dangerous.
- Final Recommendation: As the lead chemical engineer, propose a set of “optimal” conditions (temperature, pressure, and concentration management) for the industrial Haber Process plant. Explain your choices, balancing the need for a high yield (equilibrium) with the practical realities of reaction rate and cost/safety.